T core – Translation Center For Oncology Research
Clinical research refers to all research carried out on humans (healthy or sick people). It focuses on improving knowledge of diseases, developing diagnostic methods and new treatments or medical devices to ensure better patient care. It is very framed and respects a precise study protocol and is only realized under certain conditions. It must:
- have the goal of increasing medical knowledge,
- be carried out by competent persons,
- take all necessary measures to protect those who lend themselves to research,
- obtain regulatory approvals and take all the necessary legal and ethical steps.
- collect the consent of those involved in research
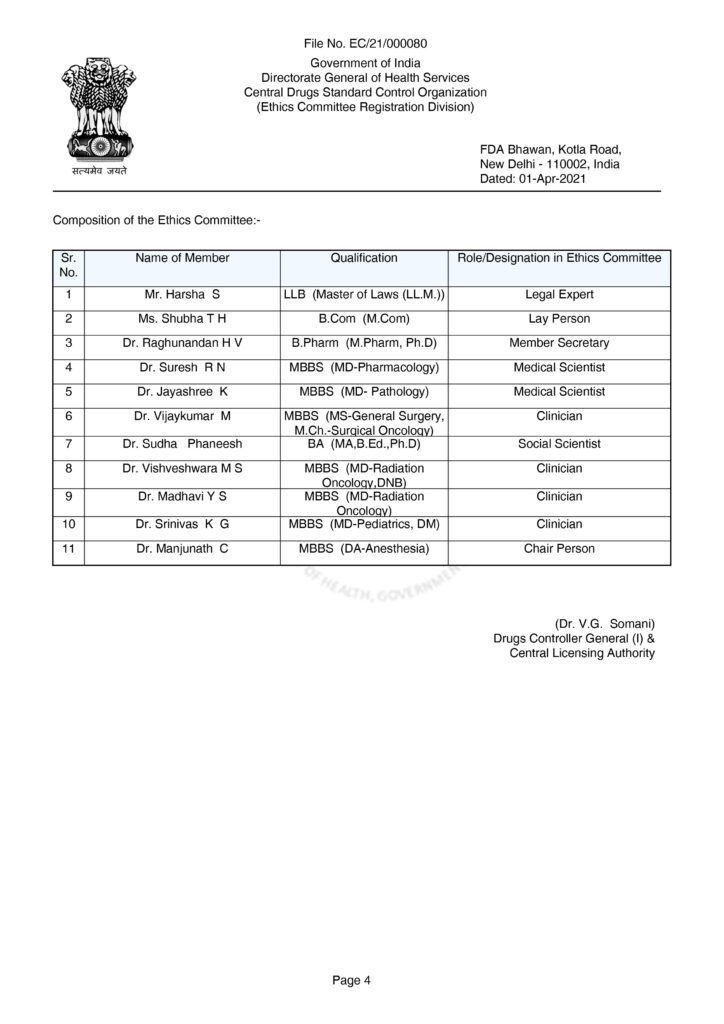
Clinical research in a hospital functions through a system of checks and balances. The Institutional Ethics Committee (IEC) provides the ethical oversight, while the Principal Investigator (PI) and Clinical Research Coordinator (CRC) handle the execution.
Institutional Ethics Committee (IEC) Roles
The IEC is an independent body responsible for protecting the rights, safety, and well-being of trial participants.
Chairman
- Role: Usually an outsider (not affiliated with the hospital) to prevent a conflict of interest.
- Responsibility: Leads meetings, ensures the independence of the committee, and signs off on final approval or rejection letters for research projects. Member Secretary
- Role: Usually an internal staff member who handles the “paperwork” and logistics.
- Responsibility: Organizes meetings, prepares agendas, maintains records/minutes, and acts as the point of contact between the researcher and the committee.
- Other Members
A diverse group ensures a balanced view:
- Medical Scientists/Clinicians: Review the science and safety.
- Legal Expert: Checks for compliance with national laws.
- Lay Person: A member of the public who ensures the study is understandable to a common person.
- Social Scientist: Evaluates the social and community impact.
The Clinical Research Team
Principal Investigator (PI)
The PI is typically a Senior Doctor/Consultant at the hospital.
- Accountability: They are legally and ethically responsible for the conduct of the trial at their site.
- Supervision: They oversee the CRC and ensure the study follows the approved protocol and “Good Clinical Practice” (GCP) guidelines.
- Medical Decisions: Only the PI (or a qualified sub-investigator) can make medical judgments, such as treating a patient for a side effect.
Clinical Research Coordinator (CRC)
The CRC is the “engine room” of the study. While the PI is responsible, the CRC does the daily work.
- Patient Management: Screens potential patients and assists with the Informed Consent process.
- Data Entry: Records all patient information into Case Report Forms (CRFs).
- Drug Accountability: Tracks the study medication—storage, dispensing to patients, and counting leftover pills.
Communication: Acts as the bridge between the Doctor (PI), the Pharma Company (Sponsor), and the Ethics Committee (IEC).
Summary of Responsibilities
Role | Primary Responsibility | Key Function |
IEC Chairman | Oversight & Bias Control | Final decision-making on ethics. |
Member Secretary | Administration | Documentation and communication. |
Principal Investigator | Clinical Leadership | Medical safety and legal accountability. |
CRC | Trial Operations | Daily management and data collection. |
IEC Chairman

Chairman – Dr. Manjunath C
Personal Details
- Name: Dr. C. Manjunath
- Date of Birth: 15.12.1948
- Gender: Male
- Nationality: Indian
Professional Experience
- Ethics Committee Experience: Working as an IEC Member for 5 years.
- Current Profession: Independent Anaesthetist Consultant
- Clinical Experience: Over 20 years of experience including roles at ,
- Mission Hospital, Mysore
- JSS Medical College
- SIMS Medical College, Shimoga
- MIMS Mandya
- Subbaiah Medical College, Shimoga
Educational Qualification
- MBBS (1976): Mysore University.
- D.A. (Diploma in Anaesthesia) (1994): Mysore University.
Contact Information
- Official Address: #438, Outer Ring Road, Hebbal, Mysuru.
- Email: drmanju15@gmail.com.
Phone: 94823 96093 / 8050477053
IEC Member Secretary

Dr. Raghunandan H V
B.Pharm M.Pharm., Ph.D
Technical Consultant–Biopharmaceuticals and Pharmaceuticals,Bharath Hospital and Institute of Oncology since 2018
Member Secretary – Institutional Ethics Committee, BHIO Unit of SSTRI, Outer Ring Road, Hebbal, Mysore -570017 since 01.04.2021
+918212300600
+919535092222
Residence address:
#1268,Srirampura II Stage I Cross Mysore -570023
Education: Registered Pharmacist
- B.Pharm., Gold Medallist and all 4years Topper 1993–MysoreUniversity
- M.Pharm., BITSPilani-1995
- Ph.D., Industrial Chemistry – Kuvempu University – 2013 Worked on formulation of gall stone treatment and Anti Depressant and Anti Obesity
Professional Affiliation
31 years Professional work experience, 17 Years in Pharmaceutical and Bio Pharmaceutical Industry in India and Abroad
1995to1996–Management Trainee – Quality Department–Cipla Limited
1996 to 2008 – GSK Positions Held – Quality Assurance Site Head, South asia Quality Senior Manager for OTC and OHC, Production and Validation Manager at GSK Philippines,
2008to2011–Biocon–Head of Quality Assurance/Senior Manager
2011 to 2018 – JSSAHER Mysore – Professor and Deputy Director Academic & Independent Quality Auditor for Pharmaceutical and Biopharmaceuticals
2018 April onwards Founder and Chief Technical Consultant at RC2 Pharma Solutions. (Pharmaceuticals and Biopharmaceuticals Regulatory and Quality) & June
2018 onwards Technical Consultant at BHIO
Technical Consultant to UL Laboratories New Jersey, USA until end 2019
Technical Consultantto United States Pharmacopoeia, Hyderabad, India Until 2022
Membership in Professional Bodies in India and a broad
General Secretary for South India Chapter of Society for Pharmaceutical Dissolution Science, Mumbai India
Past Member of ISPE–International Society of Pharmaceutical Engineers, Tampa Florida
Adjunct Professor at JSSAHER Mysore, Karnataka,
Adjunct Professor at Acharya BM Reddy college of Pharmacy Bangalore, Karnataka Until 2021
Adjunct Professor at MCOPs, Karnataka Until October 2023
Research Area work done till date
Formulation development and characterization of Anti diabetic/ Gall Stone Treatment / Anti Obsess/ Anti depressant Drugs
Quality and Regulatory work in the area of Antidiabetic/ Gall Stone Treatment / Anti Obsess/ Anti depressant Drugs
Not of Standard drugs review and policy drafting, Orphan Drugs, Irrational Drug combination/Fixed Dose combination and Regulatory policy.BA/BE studies and Clinical Trials Phase II/III/IV of anti cancer, oral Insulin, many other anti diabetic
Chemistry and Manufacturing Controls preparation for dossier, IND, ANDA and NDA document review
15 Scientific Publication available in the Indian and International peer reviewed journals
The Following Studies are Approved by the Institutional Ethics Committee are, Phase 3 trials
Phase 3 is sometimes written as phase III. These trials compare new treatments with the best currently available treatment (the standard treatment).
Phase 3 trials aim to find out:
which treatment works better for a particular type of cancer
more about the side effects
how the treatment affects people’s quality of life
They may compare standard treatment with:
a completely new treatment
different doses of the same treatment
having the same treatment more, or less, often
a new way of giving a standard treatment, such as radiotherapy
Phase 3 trials usually involve many more patients than phase 1 or 2. This is because differences in success rates may be small. So, the trial needs many patients to be able to show the difference.
Sometimes phase 3 trials involve thousands of people in many different hospitals and even different countries. Most phase 3 trials are randomised. This means the people taking part are put into treatment groups at random.
Phase 4 trials
Phase 4 is sometimes written as phase IV. These trials are done after a drug has been shown to work and has been licenced.
Phase 4 trials aim to find out:
- more about the side effects including the rarer side effects and safety of the drug
- what the long term risks and benefits are
- how well the drug works when it’s used more widely for people not included in the phase 3 trial
Bhio has complete Capability to conduct Clinical Trials in Phase 3 & Phase 4 for various Oncology related Indications
Ongoing Studies
Sl No | Company Name | Study Name |
01 | Astrazeneca | DESTINY-09-Her2+Breast Cancer (since June 2021) |
02 | Astrazeneca | CAPITello-292-Her2 Negative Breast Cancer (since May 2024) |
03 | Astrazeneca | TROPION -5-Triple Negative Breast Cancer (since May 2024) |
04 | Astrazeneca | Artemide – Biliary Tract Cancer (since Sep 2024) |
05 | Astrazeneca | CAMBRIA-2-ER+/HER2- breast Cancer (since Nov 2024) |
06 | Astrazeneca | EVOLVE LUNG-2 Non Small Cell Lung Cancer (since Jan 2025) |
07 | IQVIA | BIO-PERTUZ-301-HER2 + breast Cancer (since Feb 2025) |
08 | Astrazeneca | Evopar Breast -01- Her 2 Negative |
09 | Parexel | Tropion Lung 15 |
10 | Roche | Real world indian study of Atezolizumab in lung cancer |
Close Out Studies
Sl No | Company Name | Study Name |
01 | PAREXEL | Follicular Lymphoma (2019) |
02 | ASTRAZENECA | NILE-Ovarian Cancer (2020) |
03 | ASTRAZENECA | CAPITello-290- Triple –ve Breast Cancer (June 2021) |
04 | ASTRAZENECA | Tropion Lung -14 |
05 | ASTRAZENECA | AEGEAN-Lung Cancer (2020) (Preparing For Close Out) |
06 | DRL | DRL_NU-01-001_NSCLC |
CLINICAL RESEARCH COORDINATOR

Dr. Maniraj M R
B pharm , Pharm D (PB)
Clinical professional currently Working as a Senior Clinical Pharmacist and Senior Clinical Research Coordinator For 5 Years at the Bharath Hospital and Institute of Oncology (BHIO) in Mysore. With an advanced academic background holding both a B.Pharm and Pharm D (PB), he bridges the gap between pharmaceutical expertise and clinical research management.

Ms.Rakshitha B V B pharm
Currently Working as a Clinical Research Coordinator For 1.4 Years at the Bharath Hospital and Institute of Oncology ( BHIO) in Mysore.

Mr. Rudra Gowdu
B pharm
Currently Working as a Clinical Research Coordinator For 1.2 Years at the Bharath Hospital and Institute of Oncology ( BHIO) in Mysore.

Mr. Nur Musharaf Ahmed
B pharm
Currently Working as a Chemo Drug Reconstitution & Clinical Research Coordinator For 1 Year at the Bharath Hospital and Institute of Oncology ( BHIO) in Mysore.

Mr. Sunil Kumar Kuruba
B pharm , M pharm (Pursing)
Currently Working as a Chemo Drug Reconstitution & Clinical Research Coordinator For 4 Months at the Bharath Hospital and Institute of Oncology ( BHIO) in Mysore.